Sharps Safety
Sharps safety is an important topic affecting everyone in the medical field. Healthcare workers who directly handle sharps are obviously at risk for sticks and cuts but other workers can also be put in harm’s way when sharps are not properly stored or disposed of.
Who does it affects?
As previously mentioned, anyone working in healthcare is at risk for a percutaneous injury. From 2002 to 2011, Public Heath England ran a national Monitoring initiative to collect information on significant occupational exposures caused by sharps injuries and published the results in the Eye of the Needle report.

What is a sharp injury?
Sharps are needles, blades and other medical equipment that could cause injury by cutting or pricking the skin.1
Sharp injuries occur when an instrument accidentally penetrates the skin; this is called a percutaneous injury. If the instrument is contaminated with blood or other bodily fluids, a risk of infection transmission exists.
When this occurs in a work context, it is called occupational exposure.
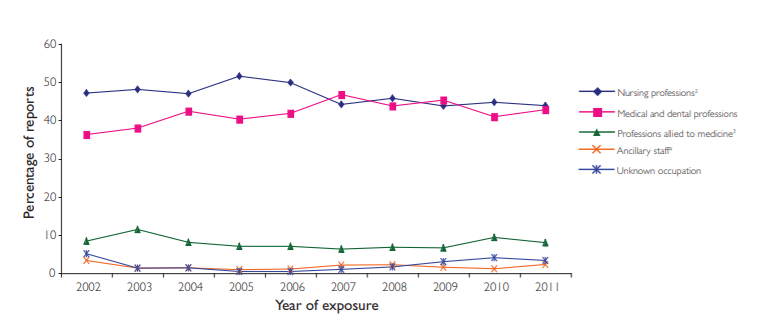
The data over the nine-year span shows that occupational exposures amongst those in nursing professions, including midwives, healthcare assistants, dental nurses and hygienists, account for almost half (46 per cent) of all reports. Medical and dental professionals follow closely behind, contributing to 42 per cent of reports.
The ancillary staff and professions allied to medicine make up roughly 9 per cent of reported occupational exposures, showing minimal change over time. Even though this is a small percentage, it is a discouraging statistic considering that 72 of these incidents involved ancillary staff, such as security and housekeeping, who are not even directly involved in patient care.2
What are some of the risks?
The main risk from a sharps injury is potential exposure to major blood-borne viruses:
- Hepatitis B virus (HBV)
- Hepatitis C virus (HCV)
- Human immunodeficiency virus (HIV)
Sharps injuries can also potentially transit the following infectious agents:
|
|
The risk of contracting an infection depends on a variety of factors, including the worker’s natural immune system, the depth of the injury, the type of sharp used, where the sharp entered the body and how infectious the patient is at the time of injury.3
In relation to the large amount of sharps injuries each year, only a small percentage cause infections that lead to serious illness. However, the effects of the injury and anxiety about its potential consequences, including the side effects of post-exposure prophylaxis, can have a significant impact on an injured employee.4
Preventing exposure
From the chief executive and the board to the healthcare staff, everybody has a role to play in sharps injury prevention. Medical professionals have a duty to protect themselves and others around them, while employers are responsible for assessing and reducing the risk of exposure as much as possible.2
The complete removal of hazardous material from the workspace is the most effective way to prevent injury, and this approach should be used whenever possible:
- Removing sharps and needles, for example by substituting jet injectors for needles and syringes or using needleless intravenous systems
- Eliminating all unnecessary injections
- Eliminating unnecessary sharps such as towel clips.
Controlling exposure
If the risk cannot be prevented, then the exposure to sharps and hazardous substances must be effectively controlled.
Implementing engineering controls, which are used to isolate or remove hazards from a workplace, can assist in significantly reducing the threat.
Some examples of these controls include the use of safety-engineered devices like retractable needles, safety lancets, blunt needles, needle counters and blade removers for all procedures.
Healthcare Legislation
Under the United Kingdom’s Health and Social Care Act, the government published a specific code of practices for the prevention and control of healthcare-associated infection. The code requires NHS bodies to implement policies that incorporate sharps protection mechanisms. The Care Quality Commission (CQC) registers and licences care services in England to ensure that they meet the fundamental standards. In regards to sharps safety, the code addresses the need to prevent the spread of blood-borne viruses by taking the following precautions:
- Immunisation against hepatitis B
- Wearing of gloves and other protective clothing
- The safe handling and disposal of sharps, including the provision of medical devices incorporating sharps protection
- Measures to reduce risks during surgical procedures.1
Related Products



